More generally, the apparition of square-like shapes on the surface of the samples after implantations and thermal treatments is discussed. The potential role played by the oxidation is underlined since even a slight modification of the surface stoichiometry may modify species mobility. Bismuth iodide perovskite solar cells materials that employ caesium as the A-site cation possesses high absorption coefficients and higher stability with. Several assumptions are proposed to explain the better retention of caesium in comparison with both other species. In comparison, iodine exhibits singular migration behaviour. Both species are homogeneously transported towards the surface and the transport rate increases with the temperature. Nevertheless, the evolution of caesium depth profile during thermal treatment presents similarities with that of xenon. 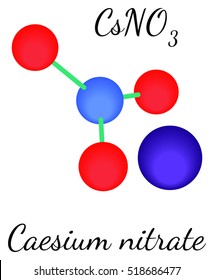
The results reveal that the global mobility of caesium in the host matrix is low compared to xenon and iodine implanted in the same conditions. Titanium nitride has been proposed as a fission product barrier in fuel structures for gas cooled fast reactor (GFR) systems. Concentration profiles were determined by 2.5 MeV 4He + elastic backscattering. Thermal treatments at temperatures ranging from 1500 to 1650 ☌ were performed under a secondary vacuum. The thermal migration of Cs was studied by implanting 800 keV 133Cs ++ ions into sintered samples of TiN at an ion fluence of 5 × 10 15 cm -2. In other words, carbonates become more thermally stable down the group.Titanium nitride has been proposed as a fission product barrier in fuel structures for gas cooled fast reactor (GFR) systems. Not for medicinal, household or other use. Relevant identified uses: For R&D use only. To compensate, the compound must be heated more in order to force the carbon dioxide to break off and leave the metal oxide. Product name: Cesium nitrate CBnumber: CB7325113 CAS: EINECS Number: 200-272-2 Synonyms: L-Glycine,Cesium nitrate Relevant identified uses of the substance or mixture and uses advised against. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and. As the positive ions get bigger down the group, they have less effect on the carbonate ions near them. Element Caesium (Cs), Group 1, Atomic Number 55, s-block, Mass 132.905. Substance information for UN 1451 - Cesium nitrate or Caesium nitrate based on the Hazardous Materials Table (Title 49 CFR 172.101) to assist in preparing a. The smaller the positive ion, the higher the charge density, and the greater the effect on the carbonate ion. The Group 1 compound must be heated more because the carbonate ion is less polarized by a singly-charged positive ion. The interaction between the support and the TMNs protects the nanoparticles from self-agglomeration and promotes charge transfer between the active site and support (Luo et al., 2016 ). This is why the Group 1 compounds are more thermally stable than those in Group 2. Mesoporous graphitic carbon nitride (g-CN) is a suitable additive with carbon black as the best performing support. If the positive ion only has one positive charge, the polarizing effect is lessened. If it is highly polarized, less heat is required than if it is only slightly polarized. The amount of heat required depends on how polarized the ion was. If this system is heated, the carbon dioxide breaks free, leaving a metal oxide. 
The diagram shows what happens with an ion from Group 2, carrying two positive charges: The positive ion attracts the delocalized electrons in the carbonate ion towards itself. Imagine that this ion is placed next to a positive ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
